Description
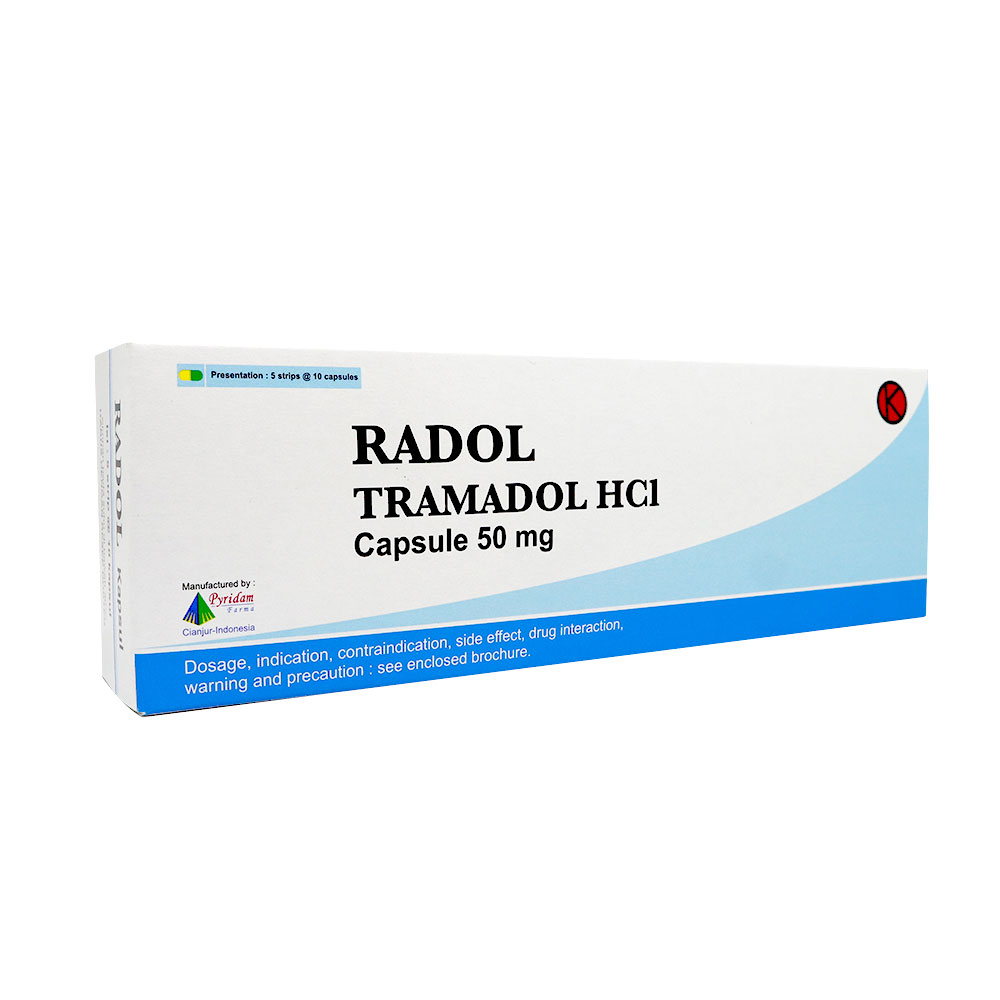
COMPOSITION :
Each capsule contains 50 mg Tramadol HCl.
ACTIONS :
Tramadol HCl belongs to the opioid analgesic group, where the potential for dependency is lower than morphine.
Opioid analgesics act on several CNS receptors which reduce moderate to severe pain.
INDICATIONS :
Severe, acute and chronic pain. Postoperative pain.
DOSAGE & ADMINISTRATION :
Adults and children > 14 years : 1 capsule as a single dose.
The dose should match the pain intensity or as advised by the physician. The previously mentioned dose is usually sufficient
to alleviate the pain.
If the pain has not lessened, an additional capsule may be taken after 30 – 60 minutes.
The dose does not depend on the mealtime.
Adjustment of the dose must be done in patients with liver and kidney dysfunction.
Total daily dose should not exceed 400 mg/day or 8 caps/day.
Long-term use : If tramadol would be used for long-term treatment, the possibility of dependence cannot be excluded.
Therefore, the physician should determine the duration of treatment or, if necessary, the treatment is discontinued temporarily.
Tramadol should not be given longer than what has been recommended.
OVERDOSAGE:
Symptoms : Myosis, vomiting, cardiovascular collapse, sedation, coma, convulsions and respiratory depression may occur.
Treatment: Convulsion can be suppressed with benzodiazepines, while respiratory depression can be neutralized by naloxone.
CONTRAINDICATIONS: Patients receiving MAOI therapy.
Acute intoxication with alcohol, hypnotics, analgesics or other agents which affect CNS.
PRECAUTIONS:
Caution should be observed in patients who are hypersensitive to tramadol or opiate.
Although tramadol is an opioid agonist, it cannot suppress morphine withdrawal symptoms.
Caution should be exercised in patients with head injury, increased intra-cranial pressure, severe liver and kidney dysfunction
or bronchial hypersecretions because it can increase the risk of convulsion and shock. Lung function may decrease if tramadol
is combined with other CNS depressants or if the recommended dosage is exceeded.
Effects on the Ability to Drive or Operate Machinery : Tramadol may affect the reaction ability of the patients, eg ability in
driving a vehicle or operating machinery.
Use in pregnancy & lactation : If tramadol is given during pregnancy and lactation, the benefits of the drug should be
considered against the risks, since 0,1% of tramadol is excreted into breast milk.
ADVERSE REACTIONS:
In normal dose, adverse reaction commonly occur which is similar to centrally acting analgesics, ie naausea, vomiting,
constipation, dyspepsia, fatigue, dizziness, pruritus, sedation, sweating, skin rash, dry mouth and headache. Although
tramadol interacts with opioid receptor, up to now, dependency after the use of tramadol is low.
INTERACTIONS:
The concurrent use of tramadol and other CNS-acting drugs, eg tranquilizers and hypnotics may enhance sedation.
Combination of tramadol with a tranquilizer may potentiate the analgesic effect.
STORAGE:
Store in a cool and dry place.
PACKING :
Box of 5×10’s. Reg.No. DKL9621011401A1.
On medical prescription only.